
Boiling, Filtering, and Distilling: Why These Are Not the Same Thing
When people start paying closer attention to their water quality, one of the first questions they ask is simple: aren’t boiling, filtering, and distilling basically the same thing?
All three involve treating water in some way, but they work very differently, and those differences matter. Understanding how each method functions, what it removes, and what it leaves behind can help households make more informed decisions about their water.
While no single method is “right” for everyone, boiling, filtering, and distilling are often confused with one another. They address very different goals.
Why Water Treatment Methods Matter
Municipal water systems are designed to deliver water that meets regulatory safety standards. These systems focus on disinfection and distribution, not on removing every dissolved substance. As a result, treated tap water can still contain minerals, disinfection byproducts, and trace contaminants — all within legal limits.
At the household level, water treatment methods typically aim to do one or more of the following:
- Kill or inactivate microorganisms
- Improve taste and odor
- Reduce or remove certain contaminants
- Standardize water quality
Boiling Water: Heat, Not Separation
Boiling water is one of the oldest and most commonly recommended emergency water treatments. When water is brought to a rolling boil, high heat inactivates many bacteria, viruses, and parasites.
What Boiling Does Well
- Inactivates many microorganisms
- Useful in short-term emergencies
- Requires no special equipment beyond heat
What Boiling Does Not Do
Boiling does not remove dissolved substances. In fact, it concentrates them.
When water boils, only the water molecules evaporate. Anything dissolved in the water — such as:
- Minerals
- Metals
- Nitrates
- Salts
- Many chemical compounds
stays behind. As water volume decreases, those substances can become more concentrated.
Boiling also does not effectively remove chlorine or chloramine, and it does not address many modern contaminants that are dissolved at the molecular level.
The Key Limitation
Boiling treats biological risk, not water composition. It is not designed to purify water in the sense of separating water from dissolved solids.
Water Filtration: Selective Reduction
Filtration is the most common household water treatment method. From pitcher filters to under-sink systems, filters work by passing water through media designed to trap or reduce certain substances.
How Filtration Works
Different filters use different technologies, including:
- Activated carbon
- Sediment screens
- Ion exchange resins
- Membranes
Each type targets specific contaminants. For example, carbon filters are commonly used to improve taste and odor by reducing chlorine.
What Filtration Can Do
- Improve taste and smell
- Reduce chlorine and some organic compounds
- Capture sediment and particulates
- Reduce select contaminants depending on design
What Filtration Cannot Guarantee
No filter removes everything. Each filter has:
- A limited list of targeted contaminants
- A finite lifespan
- Performance that depends on water quality and maintenance
Filters can also allow dissolved minerals, salts, and many small molecules to pass through unchanged. Some contaminants are simply too small or not chemically compatible with the filter media.
The Key Limitation
Filtration is selective, not absolute. It reduces what it is designed to reduce — and leaves the rest.
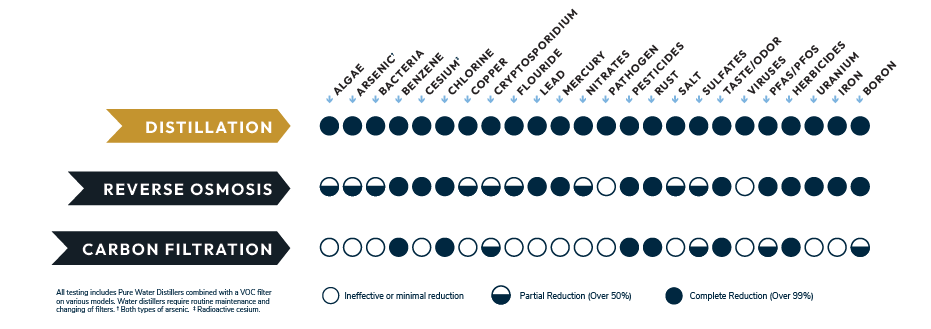
Distillation: Separation Through Phase Change
Distillation works differently from both boiling and filtering. Instead of targeting specific contaminants or relying solely on heat, distillation separates water from dissolved substances through evaporation and condensation.
How Distillation Works
- Water is heated until it becomes vapor
- Dissolved solids and most non-volatile substances are left behind
- The vapor is cooled and condensed back into liquid water
This process mirrors the natural water cycle — evaporation, condensation, and collection.
What Distillation Removes
Because most contaminants do not evaporate at the same temperature as water, distillation is effective at separating water from:
- Dissolved minerals and salts
- Metals
- Nitrates
- Many chemical residues
- Particulates and sediment
Many distillation systems also include a carbon post-filter to address volatile compounds that may vaporize along with water.
What Makes Distillation Different
Distillation does not rely on trapping contaminants or neutralizing them. Instead, it physically separates water from dissolved substances.
The result is water with a highly consistent composition, regardless of variations in source water.
Why These Methods Are Often Confused
Boiling, filtering, and distilling all involve treating water, so they are frequently grouped together. However, they serve different purposes:
| Method | Primary Function | Main Limitation |
| Boiling | Inactivates microbes | Does not remove dissolved substances |
| Filtering | Reduces select contaminants | Leaves untargeted substances |
| Distilling | Separates water from dissolved solids | Slower process |
Confusion often arises when boiling is assumed to “purify” water or when filtration is assumed to remove everything. In reality, purification depends on the goal: microbial safety, taste improvement, or total separation.
Why Consistency Matters in Water
One of the challenges with most household water treatment methods is variability. Water quality can change based on:
- Seasonal runoff
- Infrastructure age
- Local source water
- Treatment methods used by utilities
Filtration performance can vary as filters age or as incoming water changes. Boiling does not address changes in dissolved content at all.
Distillation, by contrast, produces water with a consistent composition each cycle. The process itself does not depend on incoming water chemistry in the same way.
Where Distilled Water Is Commonly Used
Because of its consistency, distilled water is widely used in settings where water composition matters:
- Laboratories
- Medical and dental equipment
- Manufacturing processes
- Appliances sensitive to mineral buildup
In homes, distilled water is often used for:
- Drinking and cooking
- Coffee and tea
- Humidifiers and steam appliances
- CPAP machines and irons
These uses are driven by purity and consistency, not health claims.
Choosing the Right Method for Your Home
Each method has a place:
- Boiling is practical for emergencies
- Filtering can improve taste and reduce certain contaminants
- Distilling provides a standardized form of water through separation
The right choice depends on what you want your water treatment system to accomplish.
For households looking to reduce variables and produce water with minimal dissolved content, distillation offers a straightforward, mechanical process based on separation rather than selective reduction.
A Simple Way to Make Distillation Part of Daily Life
Countertop and built-in water distillers allow households to distill water at home, using a process that has been relied on for decades in professional settings. Stainless steel construction, controlled heating, and carbon post-filtration help produce consistent results batch after batch.
At MyPureWater, our distillers bring the same separation-based process into everyday homes — without relying on disposable filters as the primary treatment method. Many customers choose distillation to gain greater control over the quality and consistency of the water they use every day.
The Takeaway
Boiling, filtering, and distilling are not interchangeable. Each addresses water in a different way:
- Boiling treats biological risk
- Filtering reduces select substances
- Distillation separates water from dissolved solids
Understanding those differences helps clarify why one method may be preferred over another depending on your goals.
For those seeking a consistent, standardized form of water created through physical separation, distillation stands apart — not as a replacement for all methods, but as a fundamentally different approach to water purification.







I am interested in a water treatment business here in NJ. I have a background in residential and commercial plumbing. I have sold and installed water treatment systems in the past here in NJ.