
What is Water Purification?
Water purification refers to the process of removing unwanted gases, solids, chemicals, viruses, or biological contaminants from water.
What is Distillation?
Distillation is a form of water purification, and it is also the most effective as it removes over 99% of contaminants.
5 Methods of Water Purification
-
Boiling: Heating water to a rolling boil for at least one minute kills bacteria, viruses, and other pathogens, making it safer to drink.
-
Filtration: This involves passing water through a filter to remove impurities and contaminants. Filters can vary in their pore size and material, targeting different types of contaminants.
-
Chlorination: Adding chlorine or chlorine compounds to water effectively disinfects it, killing bacteria and viruses. This method is widely used in municipal water treatment.
-
Reverse Osmosis: Water is forced through a semi-permeable membrane that filters out most contaminants, including particles, bacteria, and chemicals. This method is effective for reducing a broad range of impurities.
- Distillation: This process involves boiling water and then condensing the steam. Distilled water is free from impurities, making it very pure. [view our 3rd party verified test results]
There are many water purification methods, many of which you already know. These methods include physical processes such as sand filters and distillation, chemical methods such as chlorine treatment, and even biological and electromagnetic methods.
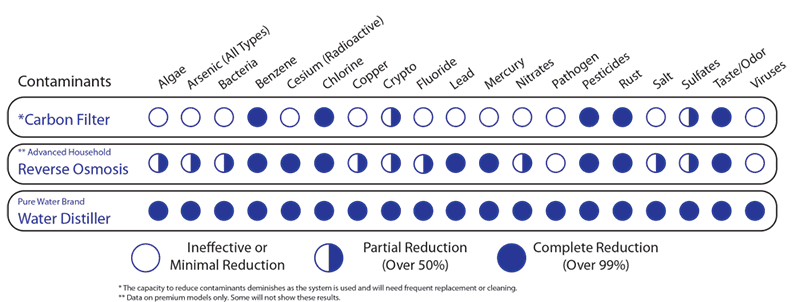
Carbon filters, reverse osmosis systems, and water distillers are the most common home water purification systems. Many opt for simple carbon filters because they are generally inexpensive. Others purchase a reverse osmosis system, thinking they remove more contaminants or make the water taste better than a simple pitcher filter. Those who have researched find that water distillers remove much more than carbon filters alone or reverse osmosis. So, what is the difference between these water purification methods, and why is distillation the best?
Carbon Filters (Filtration)
Carbon filters are made up of carbon, usually derived from coconut shells or coal. These carbon sources are ‘activated’ using heat or chemicals to create giant pores in the carbon. These porous chunks of carbon are then placed into a filter chamber, and a binding agent is added to keep the carbon in place. As the water flows through the carbon filter, the pores created in the activation process absorb contaminants and make water suitable to drink.
Carbon filters can remove numerous contaminants such as phosphates, chlorine, pharmaceuticals, pesticides, and lithium, to name a few. However, carbon filters are only effective up to a certain point and are not effective at removing certain deadly compounds such as nitrates and arsenic, of which carbon filters can only reduce up to 70%.
Reverse-Osmosis (RO) Filters
To remove more tricky contaminants such as arsenic and nitrates, reverse osmosis or distillation is necessary. Reverse osmosis works by forcing the water through a membrane with pores even smaller than those involved in a carbon filter. These pores are so small, they are able to allow the hydrogen and oxygen molecules contained in the water (H2O) through the membrane without even the smallest ion contaminants, such as nitrates.
Reverse osmosis is effective at removing an even wider range of contaminants than carbon filters. However, the efficiency of reverse osmosis filters is largely dependent on the amount of contaminant contained in the water you are trying to filter and how often you replace the membranes. For example, if your water has 30 mg/L of nitrate, reverse osmosis would be able to reduce that to 3.5-2.5 mg/L. However, if your water contains 100 mg/L of nitrates, reverse osmosis would only be able to reduce the amount of nitrates to 15-10 mg/L. This means that while reverse osmosis is more effective than carbon filters at removing certain, more dangerous contaminants, it is only effective on some amounts of these contaminants. A water distillation system will remove 99%+ contaminants known and unknown contaminants from your tap or well water.
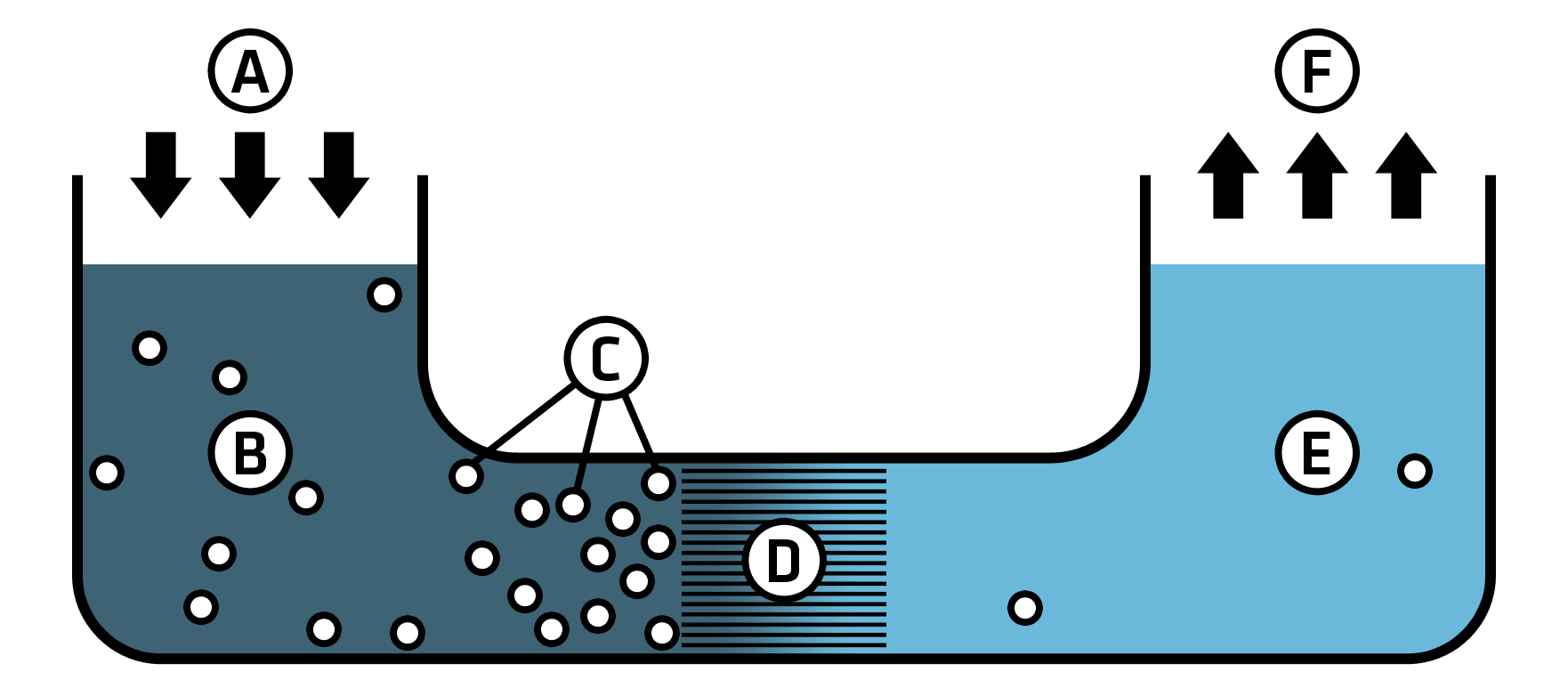
Picture source: https://commons.wikimedia.org/wiki/File:Simple_RO_schematic.png
A – Applied pressure
B – Sourcewater
C – Contaminants
D – Semi-permeable membrane
E – Potable water out
F – Distribution
Water Distillers
To remove nearly all contaminants in your water, you need a water distiller. A water distiller works by mirroring the Earth’s natural hydrologic cycle, which is how the Earth creates rain through evaporation. First, heat from the sun evaporates water from the surface, bringing it into the atmosphere. Under colder temperatures, the water is condensed and eventually falls to the ground. A water distiller works the same way.
First, heat is applied to the boiling tank’s source water until it becomes steam. This steam is then condensed and collected in a tank or jar for drinking. Because the water is boiled and only its steam is collected, virtually every contaminant contained in the source water is left in the boiling chamber. The result is 99% pure water! A water distiller can remove arsenic, nitrates, lithium, pesticides, fertilizer, pharmaceuticals, and nearly anything else you can think of, even uranium.
As shown in the video below, even if the water is dyed red, the water comes out perfectly clear.
What’s the Best Drinking Water?
Although a water distiller is initially more expensive than a carbon filter, or a reverse osmosis system, it’s the most effective at purifying drinking water. While distillation can remove over 99% of contaminants, carbon and reverse osmosis filters can only reduce some of contaminants in your water.
A water distiller is the best way to purify water. Pure Water Distillers come in manual fill countertop units and fully automatic units that you can connect to your refrigerator or kitchen sink. Pure Water distillers also have the industry’s best warranty and last many years. Enjoy high-quality, refreshingly delicious water with a Pure Water Distiller.





Can the distillation system remove microplastics? and hormones?
What sort of hormones do you have high concentrations of? If the vaporization temp of said hormones is higher than 212 degrees F they will be left with the residual source water at the end of the distillation cycle.
What is the measurement of TDS (total dissolved solids) and Electrical conductivity of your distilled water that comes out of the mini classic distiller
With the Mini Classic, there is room for human error but you should expect results between 0-7 ppm TDS. EC will also be between 1-5 mS.
If you could let me know about the mineralisation of the water. I have heard that distilled water leaches the minerals out of the body because there are no minerals in the water. That is why I have never used distilled water. I have also heard that people mineralise their reverse osmosis and distilled water because of this issue.
People say the distilled water is detrimental to the health.
This leaves me confused are you able to help me with this?
thank you
Rhonda
Rhonda, this is a great and common question we get! Although it is a common misconception in the drinking water industry, no type of water has the ability to leach minerals from your body. We know this because of how water and minerals get absorbed in the body in the small intestines. Water is absorbed into areas of high concentrations of minerals, while minerals area either brought in, or diffuse into areas with lower concentration of that mineral. After nutrients are absorbed, they remain in the walls of the small intestine and are not let out unless there is an excess and the body purposely removes it by excretion or urine. This is why it is not possible for water to leach or reduce the total number of minerals in your body simply by being too pure. It is possible however to drink too much water and reduce the proportion of water to minerals in your body, which is why it is always good to keep a healthy, nutritious diet along with the water you drink.
However, if you are interested in having minerals in your water, it’s always fine to add them back in after distillation. This way, at least you know that there is absolutely nothing in your water other than H2O and the minerals you added in. Just make sure the minerals you add are in a chelated, bioavailable form so you get the best bang for your buck!
We hope this helps!
Here is a great resource to learn more about how the small intestines absorb minerals and water: https://rabowen.org/hbooks/pathphys/digestion/smallgut/index.html
There is advise about making sure you eat a nutritional diet that provides all the minerals your body needs. This way you will get all the nutritions you need while using distilled water as your main water source
My immediate family and I have been using distilled water for drinking and food preparation since 1974; in fact, I was a distributor for Pure Water, Inc. for many years.
In the company presentation in this article nothing was said about the fact that distilled water does not conduct electricity. What I have left over from my days of selling people on the need or necessity of consuming only distilled water, I have a tester for distilled water. It is a plastic tube approximately 5/8″ diameter x 5″ long with two wires separated at the ends and connected to a cable about 4 feet long with a plug to connect to an AC electric outlet. We buy a lot of our needs from Walmart, so when I noticed that Walmart sold Gallon jugs of distilled water, I bought one to test for purity with my electric tester. Their so called distilled water was not distilled water. It may have been RO or worse. I am acquainted with the manager of our Walmart store, so next time I saw him, I told him that his store was not honest about distilled water. His answer was expected- one small voice among many.